How are ice core dated?
- Florent Fayolle
- Dec 9, 2018
- 2 min read
Ice core dating is an important part of studying items that it contains such as black carbon and trapped gas.
In this post, I decided to lighten the main methods used to date ice cores.
Counting layers
This method is based on finding and looking at seasonable depend markers trapped within the ice. Those markers can be defined within a layer of ice core, and counting those layers allows us to determine the period of time that the ice core accumulated over.
These analysed markers need to be temperature-dependent.
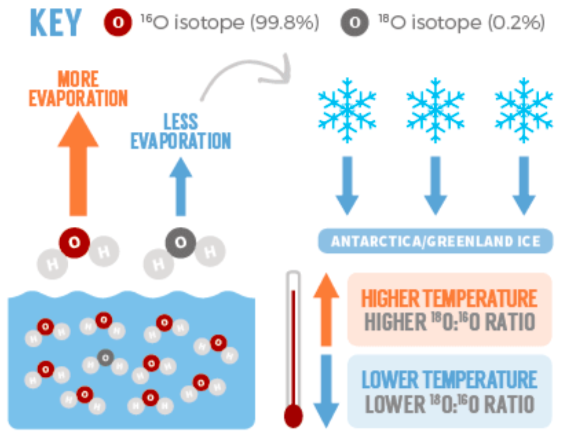
Isotopes of oxygen rely on temperature variations:
Isotope O18 are heavier than isotope O16 so are less affected by evaporation than isotope O16. During wintertime, low evaporation comes with ice cores depleted in O18 and enriched in O16. At summertime, high evaporation transports more O18 to the poles, resulting in more O18 enriched ice cores. ( See picture on the side)
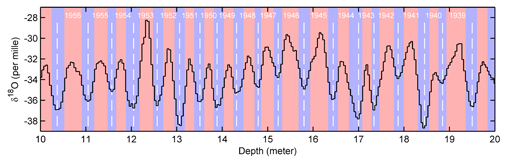
The picture above shows the variations in isotope O18 in an ice core and the delimitations of annual years. Annual layers are determined by analysing the concentration in isotope O18: then a one year layer typically starts with a low concentration of O18 (winter), then the high concentration in O18 (summer) and finally low concentration of O18 starting October. Finally, the number of layers is calculated and a time scale for the ice core can be found. An example can be found with the study of an ice core from the Vostok glacier.
Use specific ages as markers
Volcanic events can be used to date ice cores as their ash is sometimes trapped and preserved in ice cores as particles.
But for that, it is necessary to first know the date of the eruption and its source. Dark impurities from volcanic origins can be trapped in the ice core as fine layers.
Radiocarbons measurements
With this method, a section of the ice core is totally melted and delivers gas that was trapped. These gas contain a little amount of Carbon and Carbon 14 measurements can be done. Carbon 14 is a radioactive isotope of the Carbon chemical element. The amount of this radioactive carbon decreases with time so it is not an accurate technique. Other problems related to this method are stated in a study at the University of Arizona.
All methods have disadvantages!
The methods above are the main ones used to date ice cores but still have limits. For instance, using isotopes to date cores can sometimes give errors as it is known that isotopes tend to diffuse over time. There are also other methods that I decide to not talk about.









Comments